| Table of Contents |  |
|
Original Article
| ||||||
| Effect of biosurfactant produced by Citrobacter murliniae AF025369 and a synthetic surfactant on degradation of crude oil | ||||||
| Chikodili Gladys Anaukwu1, Chinyere Constance Ezemba2, Vivian Nonye Anakwenze1, Kingsley Chukwuebuka Agu1, Benjamin Chidi Okeke1, Nsikak Sunday Awah1, Ikechukwu Amaechi Ekwealor1 | ||||||
|
1Department of Applied Microbiology, Nnamdi Azikiwe University, Awka, Anambra state, Nigeria.
2Department of Microbiology, Rennaissance University, Enugu, Nigeria. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Anaukwu CG, Ezemba CC, Anakwenze VN, Agu KC, Okeke BC, Awah NS, Ekwealor IA. Effect of biosurfactant produced by Citrobacter murliniae AF025369 and a synthetic surfactant on degradation of crude oil. Edorium J Microbiol 2016;2:1–6. |
|
Abstract
|
|
Aims:
To compare crude oil degradation by Citrobacter murliniae AF025369 in the presence of biosurfactant produced by Citrobacter murliniae AF025369 and a named chemical surfactant.
Methods: Biosurfactant-producing bacterium was isolated from spent engine-oil polluted soil and analyzed for biosurfactant production in mineral salt medium using 2% glycerol as sole carbon source. The isolate was identified based on 16S rDNA sequence. Biosurfactant production was determined by drop collapse test, oil displacement test and emulsification index measurement. Tensoactive property and bioemulsification activity of the biosurfactant was carried out on various hydrophobic substrates. Crude oil degradation was investigated by gravimetric analysis. Results: The biosurfactant production assay gave an emulsification index (E24) of 66.67%, oil displacement of 1.8 cm and a positive drop collapse. It reduced surface tension of water from 72–42 mN/m, with critical micelle concentration (CMC) of 60 mg/L. Assessment of bioemulsification activities of the biosurfactant produced by C. murliniae with various hydrocarbon substrates gave highest emulsification index (E24) of 73.33% with palm oil, 70% E24 with crude oil and least E24 of 33.33% with fuel. Crude oil degradation analysis revealed that C. murliniae was able to degrade crude oil by 94%, it attained 96% crude oil degradation when the biosurfactant was introduced into the medium and 78% degradation when supplemented with Tween-80. Statistical analysis indicates that there is significant difference on the degradation rate using one-way ANOVA (p-value 0.0004). Conclusion: The results obtained show that biosurfactant was a better biostimulant and has great potential to be used in bioremediation processes, especially in the petroleum industry. | |
|
Keywords:
Biosurfactant, Citrobacter murliniae, Crude oil degradation, Emulsification index
| |
|
Introduction
| ||||||
|
Surfactants (surface active agents) are amphipathic molecules with both hydrophilic and hydrophobic (generally hydrocarbon) moieties that partition preferentially at the interface between fluid phases with different degrees of polarity and hydrogen bonding such as oil/water or air/water interfaces [1]. Such characteristics confer excellent detergency, emulsifying, foaming, and dispersing traits, which make surfactants one of the most versatile process chemicals [2]. Biosurfactants are surface active agents of microbial origin. Biosurfactants are molecules that exhibit pronounced surface and emulsifying activities, produced by a variety of microorganisms [3]. Biosurfactants are mainly categorized by their chemical composition and microbial origin [4]. Biosurfactants are classified based on chemical nature into glycolipids, lipopeptides, lipoproteins, phospholipid, fatty acids, polymeric biosurfactant and particulate biosurfactants [5]. The enormous market demands for surfactants are currently met by numerous synthetic mainly petroleum based, chemical surfactants. These compounds are usually toxic to the environment and non-degradable [6]. A host of interesting features of biosurfactants such as high biodegradability, low toxicity, and effectiveness at extremes of temperature, pH and salinity have led to a wide range of potential applications in the oil recovery, environmental bioremediation, food processing and medicine fields [3]. Biosurfactants are considered secondary metabolites, however, they confer a wide variety of roles for the survival of the producing organisms such as facilitation of nutrient transport across the membrane, microbe-host interaction and inhibitory activity against pathogenic organisms [7][8]. Many reviews have summarized the possible roles of biosurfactants [9][10][11]. These include increasing the surface area and bioavailability of hydrophobic water-insoluble substrates, heavy metal binding, bacterial pathogenesis, biofilm formation and quorum sensing [9]. The largest possible market for biosurfactant is the oil industry, both for enhanced oil recovery and for incorporation into oil formulation [12]. Hydrocarbon degrading and surfactant producing bacteria belong to the genera Pseudomonas, Achromobacter, Flavobacterium, Micrococcus, Bacillus, Arthrobacter, Klebsiella, Acinetobacter, Aeromonas, Alcaligenes, Streptococcus, Corynebacterium, Moraxella, and Proteobacteria [13]. | ||||||
|
Materials and Methods
| ||||||
|
Microorganism Inoculum preparation Fermentation Biosurfactant production assay Drop collapse method of screening The oil displacement technique Emulsification index Surface tension measurement The concentration at which micelles began to form was represented as the Critical micelle concentration. The CMC value was determined by plotting the surface tension as a function of the biosurfactant concentration Assessment of emulsifying activity Purification of biosurfactant Crude Oil Degradation Extraction of Crude Oil The amount of residual oil was measured after extraction of oil from the medium and evaporating it to dryness. The percentage of degradation of oil was calculated as follows: Weight of Residual crude oil= Weight of beaker containing extracted crude oil – Weight of empty beaker. Amount of crude oil degraded = Weight of crude oil added in the media –Weight of residual crude oil %degradation = (Amount of crude oil degraded/Amount of crude oil added in the media) x100 Statistical analysis | ||||||
|
Results | ||||||
|
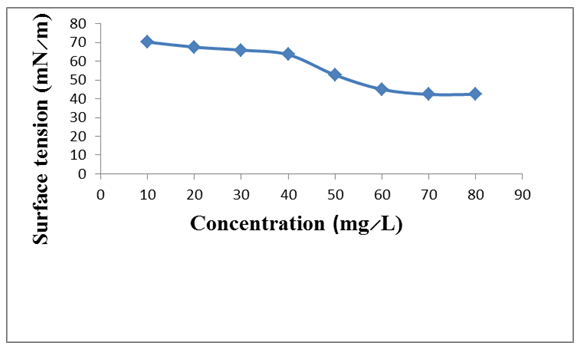
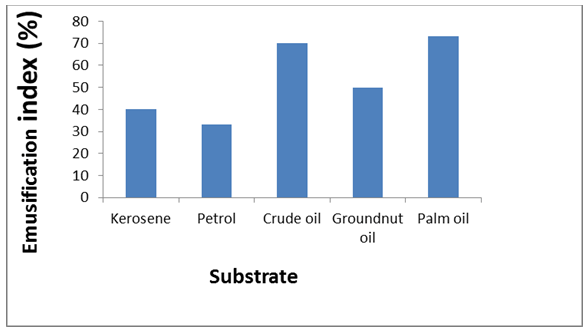
The biosurfactant production assay showed bacterial growth of 1.102. Biosurfactant produced by Citrobacter murliniae AF025369 had emulsification index of 66.67%, maximum oil displacement of 1.8 cm and a positive drop collapse (Table 1). The biosurfactant produced reduced surface tension of water from 72–42 mN/m, with critical micelle concentration (CMC) of 60 mg/L (Figure 1). The bioemulsification assay is shown in Figure 2. Highest emulsification was obtained with palm oil substrate at E24 of 73.33%, followed by crude oil at E24 of 70%. Petrol gave least emulsification (E24 = 33.33%). Crude oil degradation analysis is given in Table 2. C. murliniae AF025369 obtained 94% degradation of crude oil. In the presence of biosurfactant, it obtained 96% degradation and least degradation rate with Tween-80. | ||||||
| ||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
Microorganisms of the class Enterobacteriaceae have been discovered to be biosurfactant producers. In this study, Citrobacter murliniae AF025369, isolated from spent engine-oil polluted soil was identified as a biosurfactant producer. This is supported by the works of Thavasi et al. [22] and Mandal et al. [23]. While Thavasi et al. [22], isolated Citrobacter intermedius alongside Klebsiella ozaenae as biosurfactant producers, Mandal et al. [23], isolated and characterized Citrobacter and Enterobacter as lipopeptide biosurfactant producer. The screening procedures used were consistent with previous works [24] [25]. In the biosurfactant production assay (Table 1), the cell free culture broth gave positive drop collapse result, 1.8 cm oil displacement diameter and 66.67% emulsification index measurement. The oil displacement diameter result is quite contrary to the works of Hesham et al. [26] and Jaysree et al. [27]. While Hesham et al. [26] obtained rate of oil displacement ranging from 2.8 cm to 4.1 cm in the screening of Candida species for biosurfactant production, Jaysree et al. [27] recorded displacement diameter ranging from 3.0 cm to 4.2 cm in their work on biosurfactant production by halophilic bacteria. Emulsification activity is one of the criteria to support the selection of potential biosurfactant producers [25] . The cell-free culture broth used in the emulsification index assay contains biosurfactant; it emulsified the kerosene present in the test solution by 66.67%. Surface tension measurement of the cell-free culture broth obtained in this study (Figure 1), showed that C. murliniae AF025369 was able to reduce surface tension of water from 72–42 mN/m. Critical micelle concentration (CMC) was reached at 60 mg/L This is indicative of the tensoactive property of the produced biosurfactant. Bioemulsification results (Figure 2), show that biosurfactant produced by the C. murliniae AF025369 formed stable emulsions with the hydrophobic substrates tested. It gave considerable emulsification activity with crude oil, thus showing potential for crude oil bioremediation. Maximum emulsification of 73.33% was obtained with palm oil substrate. Thavasi et al. [22], in their study, observed that the biosurfactant produced by Pseudomonas aeruginosa was able to emulsify several hydrophobic substrate (waste motor lubricant oil, crude oil, peanut oil, kerosene, diesel, xylene, naphthalene and anthracene) better than the synthetic surfactant tested. Result on crude oil degradation (Table 2), shows that Citrobacter murliniae AF025369 is a crude oil-degrading bacterium. It utilized the crude oil as source of energy. Although C. murliniae AF025369 showed maximum degradation in the presence of biosurfactant. It also showed high degrading capacity in the absence of any stimulation by surfactant. Comparatively, biosurfactant produced by C. murliniae AF025369 stimulated its crude oil degradation potential than Tween-80. The organism without the biosurfactant also performed optimally than in the presence of the chemical surfactant. This could be that the chemical surfactant had inhibitory effect on the organism. | ||||||
|
Conclusion
| ||||||
|
From the results obtained in this study, Citrobacter murliniae AF025369 is a crude oil-degrading bacterium. The biosurfactant produced had low critical micelle concentration (CMC). The emulsification study showed that Citrobacter murliniae AF025369 can utilize varieties of hydrophobic substrate as energy source. The biosurfactant produced performed better than synthetic surfactant in crude oil biodegradation. The overall results suggest that C. murliniae AF025369 is a good producer of biosurfactant which possess potential for biostimulation in crude oil bioremediation. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Chikodili Gladys Anaukwu – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, final approval of the version to be published Chinyere Constance Ezemba – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Vivian Nonye Anakwenze – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intel0lectual content, Final approval of the version to be published Kingsley Chukwuebuka Agu – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Nsikak Sunday Awah – Ssubstantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Benjamin Chidi Okeke – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Ikechukwu Amechi Ekwealor – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Chikodili Gladys Anaukwu et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|