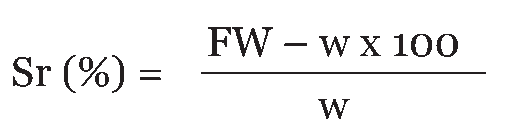| Table of Contents |  |
|
Original Article
| ||||||
| Evaluation of a new method for testing the pathogenicity of molds to yam tubers | ||||||
| Frank Chukwunwike Ogbo1, Kingsley Chukwuebuka Agu2 | ||||||
|
1PhD, Ogbo Frank C- Professor, Applied Microbiology and Brewing Department, Nnamdi Azikiwe University, PMB 5025, Awka, Anambra State, Nigeria.
2MSc, Agu, Kingsley C- Lecturer II, Applied Microbiology and Brewing Department, Nnamdi Azikiwe University, PMB 5025, Awka, Anambra State, Nigeria. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Ogbo FC, Agu KC. Evaluation of a new method for testing the pathogenicity of molds to yam tubers. Edorium J Microbiol 2015;1:9–17. |
|
Abstract
|
|
Aims:
The Objective of this study was to compare methods involving cutting of yam, with a method developed in our lab, which exposes the yams to the pathogens but without cutting the protective skin as well as to estimate the weight loss and percentage severity of rots caused by rot-causing molds of yam tubers.
Methods: A total of thirteen test molds viz., Aspergillus sp., Aspergillus tamarii, Aspergillus niger, Aspergillus flavus, Fusarium solani, Lasiodiplodia theobromae, Mucor circinelloides, Paecilomyces sp., Fonsecaea sp., Phialophora sp., Graphium sp., Saccharomyces sp. and Exophiala sp., as identified using the partial ITS rDNA sequencing analysis and a BLAST search using the GenBank sequence database, isolated from previous works from five yam varieties namely: Dioscorea dumetorum, two varieties each of D. alata and D. rotundata, were used for this study. Comparative pathogenicity tests were carried out with fresh, healthy yam tubers by the methods described by Okigbo and Nmeka, (2005) and a method developed in our laboratory. The percentage weight loss over a period of four weeks, symptoms observed and percentage severity of rots produced by the pathogenic molds were all studied. Results: The ability of both tests to detect severity of spoilage were studied and recorded as + (symptoms noticed), – (No symptom noticed) and +/C (symptoms noticed and consistent). Only seven isolates were pathogenic by the method of Ogbo and Agu, whereas all thirteen isolates were pathogenic by the method of Okigbo and Nmeka (2005). There was no significant difference p>0.05 between the weight losses and percentage severity of rots amongst the test molds with both methods except in the case of D. alata var. abana mmee tested with Fusarium solani which displayed high percentage rot severity of 450.00 by the method of Ogbo and Agu as against 153.85 displayed by the method of Okigbo and Nmeka (2005). Results obtained helped to ascertain the state of the molds (opportunistic pathogens or pathogens). Conclusion: Only molds capable of breaching the protective cover of yam tubers and eliciting original symptoms consistent with those seen in previous studies and reported in other literatures were actual yam pathogens. | |
|
Keywords:
Fungal rots, Fungal spoilage, Pathogenicity tests, Yam varieties
| |
|
Introduction
| ||||||
|
Yams (Dioscorea spp.) are perennial herbaceous vines grown for the consumption of their starchy tubers in West Africa, Asia, Carribean, Latin-America, Oceania islands of the South Pacific and some parts of Brazil [1]. Out of the World production of 50 million tonnes of yams per annum, West and Central Africa account for about 94% of the world's production, with Nigeria ranking highest by producing 35 million tonnes [2][3]. In spite of this, the demand of yam tubers in Nigeria has always exceeded its supply as a result of post-harvest losses emanating from fungal rots. After poverty per se, the third world's most burning challenge is food scarcity. The International Food Policy Research Institute [4] predicted a dismal future for the world's food condition. Post-harvest storage loss is one of the important sources of food insecurity in West Africa and is a major concern even to the United Nations which brought it to international focus when it announced in 1975 that "further reduction of post-harvest food losses in developing countries should be undertaken as a matter of priority" [5] [6]. In Nigeria, post-harvest spoilage of yam tubers ensuing in microbially-induced rot is one of the principal influences affecting commercial production of yam [7]. Various fungi have been recognized by several researchers as causative agents of numerous yam diseases including, Colletotrichum gloeosporioides as a fungal pathogen infecting minisettes through infested tubers. Botryodiplodia theobromae, Aspergillus niger, A. tamarii, Cladosporium herbarum, Cylindrocarpon radicola, Gliocladium roseum, Geotrichum candidum, Gliomastix convoluta, Macrophomina phaseoli, Mucor circinelloides, Fusarium moniliforme, F. solani, Penicillium cyclopium, P. sclerotigenum, Rhizoctonia solani, Rhizopus nigricans, R. stolonifer, and Rosellinia spp. and other fungal pathogens associated with rots of yam tubers [8] [9] [10] [11] [12]. There is a need to have precise tests to determine pathogenicity of those mold species and their roles in yam spoilage. Knowledge will help develop sufficient preservation methods. Slices of potato tubers arranged on a filter paper placed inside a Petri dish and subsequent inoculation of fungal spores have been used to assay for the pathogenicity of Fusarium isolates [13] whereas, the boring of holes of various sizes in healthy yam tubers and incorporation of several days old fungal cultures into the holes and subsequent replacement of the removed tissue and sealing with petroleum jelly have been performed by various researchers including [14] [15] [16] [17]. However, it is well known that the protective cover of foods play a major role in the prevention of microbial invasion and subsequent attack. The aim of this work, therefore, is to compare methods involving cutting of yam, with a method developed in our lab, which exposes the yams to the pathogens but without cutting the protective skin. Results which reveal pathogens with ability to spoil intact yam tubers would be accepted. | ||||||
|
Materials and Methods
| ||||||
|
Yam Collection and Identification Test Molds Pathogenicity Studies
The developing symptoms and rate of spoilage were monitored weekly for a period of one month and recorded. Yam tuber impregnated with only agar was also set up as the control and monitored weekly for a period of one month [14]. A second method developed in our laboratory involved obtaining five-day old pure cultures of the suspected rot-causing fungi and transferring them in sterile bandages. Thereafter, the culture-bandage arrangements were used to dress selected healthy yam tubers that were surface sterilized and having nearly similar weights with those used for the previous study. A control containing only bandage and un-inoculated agar was also set up. The yam tubers were then stored in a yam barn until rots developed. Observations about rate of decay and weight loss were also recorded weekly for a period of one month. The results obtained from the two methods were matched with those obtained from a previous study "Role of Fungal Rots in Post-harvest Storage Losses in Some Nigerian Varieties of Dioscorea Species" (Ogbo and Agu). Symptoms of Yam Rot Hand feel and visual examination, followed by examination of the interior of suggestive tubers was used to ascertain the symptoms of tuber rots. Rots were categorized using the scheme of Amusa et al. [18] as follows: Dry rot: The infected tissues became hard and dry with varying discolorations depending on the causative agent. Soft rot: The infected tissues became soft and ramified by the fungal mycelium. Wet rot: This was typified by the exudation of whitish fluid from the infected yam tissue when pressed between the fingers. Percentage Severity of Rots The severity of rot seeks to measure the magnitude of the infection as well as the rate of the pathogenicity of the rot-causing fungi. This was determined by obtaining the rotted portions off the whole tubers and taking the final weight of the individual yam tuber. The percentage severity of rot (Sr %) was calculated thus: | ||||||
| ||||||
|
where, FW = Final weight of infected yam tuber, w = weight of rotted tuber portion. Determination of Percentage Weight Loss Weight loss (%) was calculated from a comparison of the weight of individual yam samples at the commencement and at the end of experiment. Statistical Analyses All measurements were carried out in triplicate and data subjected to Analyses of Variance (ANOVA) using IBM SPSS Statistics version 20. | ||||||
|
Results | ||||||
|
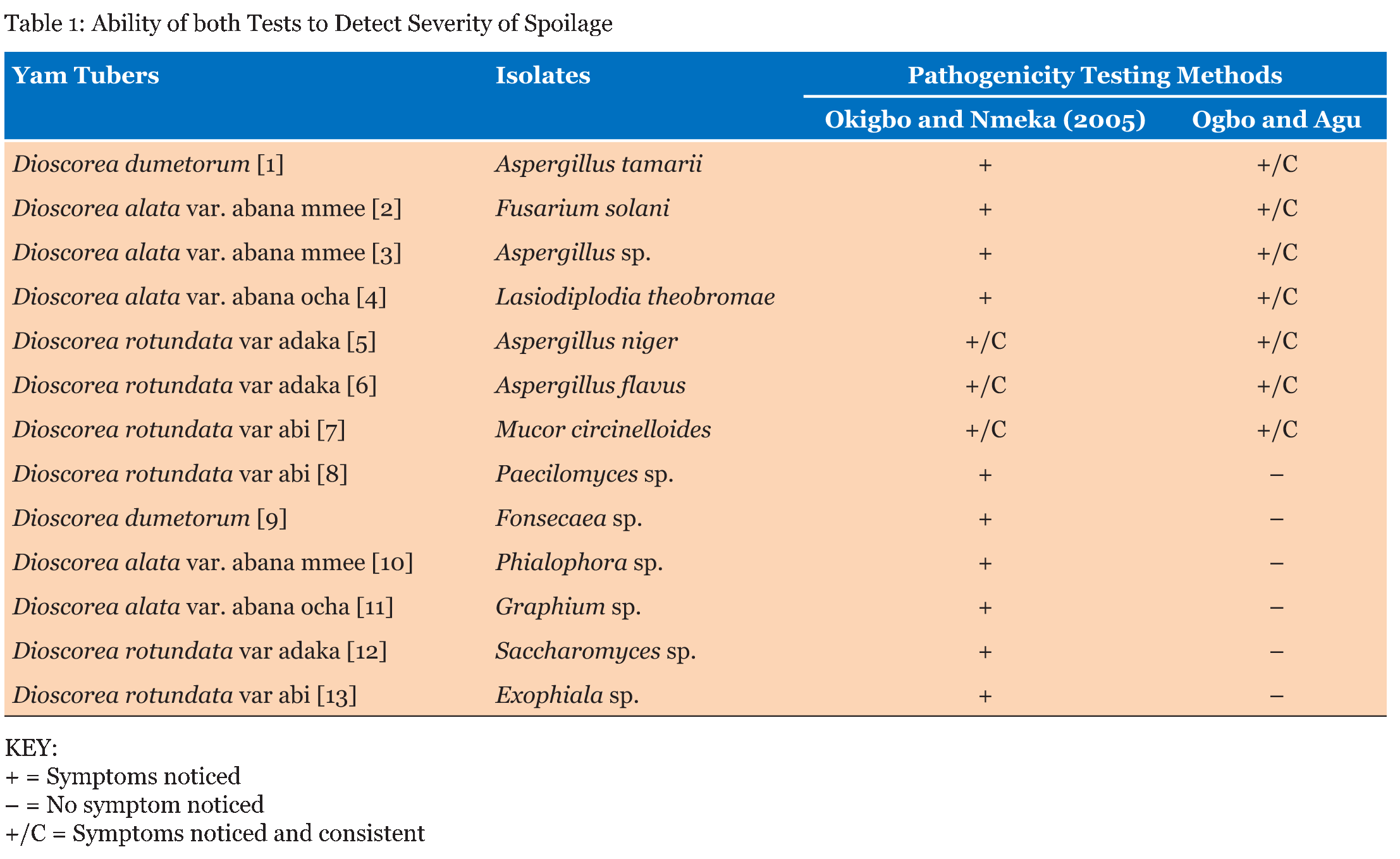
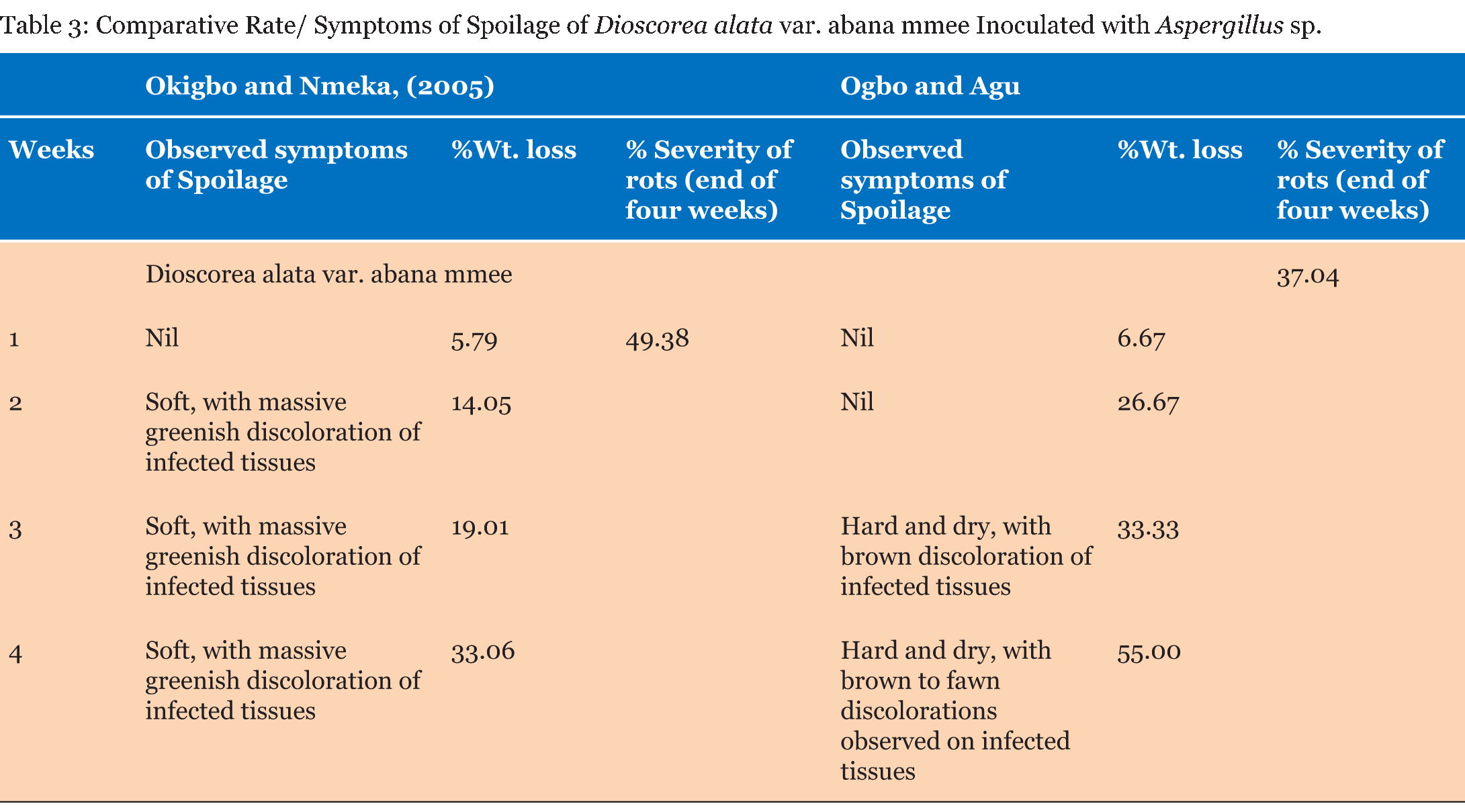
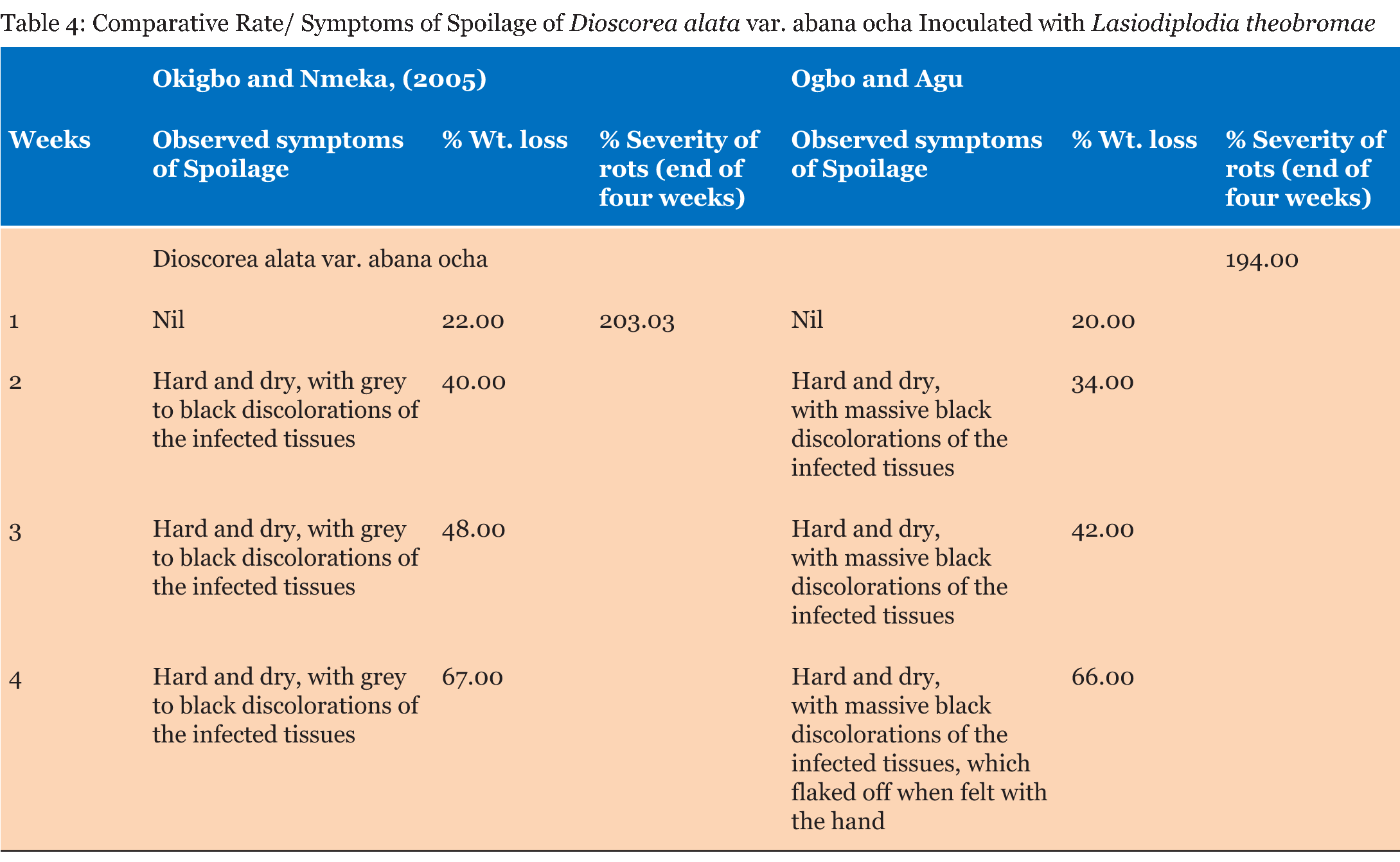
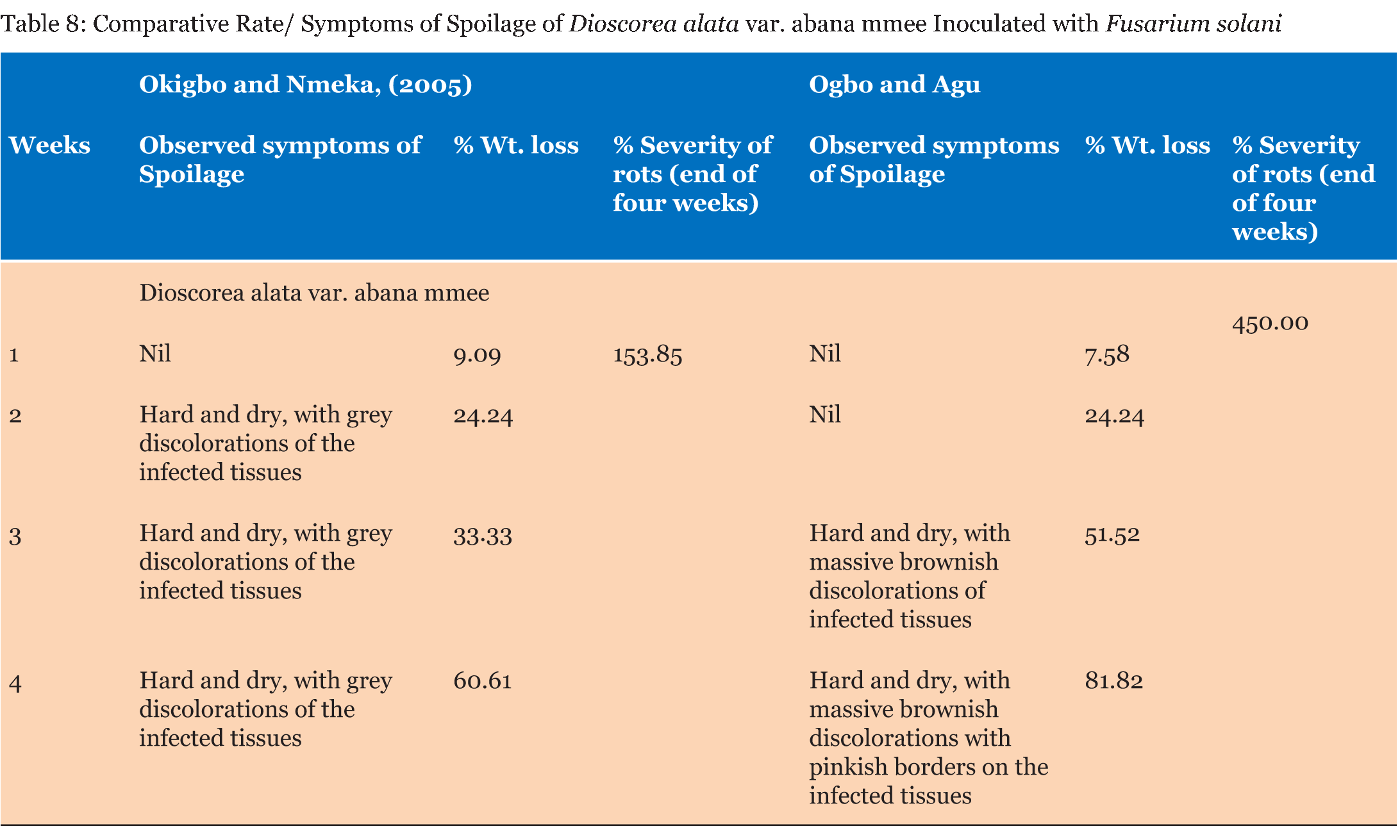
Test tubers were observed for developing symptoms and reported as +, – or +/C. There was no significant difference in the percentage weight loss and percentage severity of rots in the yam tubers tested by both methods as seen in (Table 1) (Table 2) (Table 3) (Table 4) (Table 5) (Table 6) (Table 7). Apart from percentage severity of rot of Fusarium solani which was significantly different in both methods as seen in Table 8. | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
The ability of both tests to detect severity of spoilage were studied and recorded as + (symptoms noticed), – (No symptom noticed) and +/C (symptoms noticed and consistent) and presented in (Table 1). Symptoms were observed in all the tubers studied by the Method of Okigbo and Nmeka (2005), but only three isolates viz. Aspergillus flavus, Aspergillus niger, and Mucor circinelloides were consistent with previous symptoms observed in the diseased yam tubers from where they were isolated. Seven out of the thirteen isolates elicited symptoms consistent with previous studies. This served as the yardstick for measuring the veracity of both pathogenicity testing methods employed in this study based simply on conformity of the symptoms observed with those reported in a previous study. The results of the symptoms observed in the former method were not in tandem with those observed in a previous study, whereas the symptoms observed in the latter method showed absolute conformity with the symptoms observed in a previous study. This could be suggestive of the fact that most of the molds that showed pathogenicity with the former method could have been secondary pathogens and were not able to out-compete the primary pathogens in establishing a disease in the presence of possible aerial contamination. The results of the Comparative pathogenicity tests using the methods of Okigbo and Nmeka, (2005) as well as the method Ogbo and Agu (Table 2) (Table 3) (Table 4) (Table 5) (Table 6) (Table 7) (Table 8) has shown that high percentage severity of rots were recorded in the former method except for D. dumetorum and D. alata var. abana mmee. This may be as a result of the ability of the pathogens to utilize the nutrients of the tubers as substrates for growth and development. This result is similar to the report on fungi associated with Nigeria yam [19]. In both methods, however, there were high percentage weight loss of inoculated tubers during the four weeks pathogenicity test period. Again, this may be due to the fact that the nutrients viz. carbohydrate, moisture, protein, fiber and fat of the tubers were being degraded by the pathogenic molds as well as depletion emanating from auto-spoilage during storage. This finding is in consonance with more recent report by Ikediobi CO and Oti E [20] and even more recent report by Frank CO and Kingsley CA [12] they showed that perhaps, the most prominent physiological change occurring in the tuber during storage is respiration. This leads to the loss of dry matter and quality of the tuber as food. Again, El-Hassan et al. [13], and Osunde ZD and Orhevba BA [21] reported that the physiological changes occurring within the tuber, which determine the shelf life of yams primarily, arise from sprouting, transpiration, which is essentially a physical evaporation process and respiration. This phenomenon is significant because among other effects, it leads to the shriveling and loss of the culinary quality of the tuber. The former method revealed that all the test fungi were pathogenic to the yam tuber samples. This may be as a result of the fact that the tubers were first wounded and the fungi inoculated into them, in conjunction with the fact that fungi are amylolytic and yam tubers are good sources of carbohydrates. Thus, even the non-pathogenic molds, became opportunistic pathogens. Owing to this limitation a second method developed in our laboratory was employed which brought the integrity of the whole tuber into focus. Thus, the second method proved to be a better method for testing the pathogenicity of fungal pathogens of yams and will help yam pathologists in various yam growing zones of the world, to establish original yam pathogens thereby boosting the overall output of yam production and minimizing post-harvest loss of yam production to fungal pathogens. This method revealed only seven out of the thirteen isolates to be pathogenic. Pathogenicity was based on the ability of isolates to be able to colonize, penetrate and elicit symptoms and possibly establish the disease it caused originally. This data fairly agrees with the findings of Amusa et al. [18] they carried out a survey of the micro organisms associated with the stored and marketed yam tubers obtained from the tropical forest region of South-western Nigeria and their pathogenicity on yams and reported that dry rot is considered as the most devastating of all the storage diseases of yam tubers as well as dry rot of yams alone causes a marked reduction in the quantity, marketable value and edible portions of tubers and those reductions are more severe in stored yams It is also in consonance with more recent reports [22] [23]. Aspergillus tamarii exhibited the highest percentage severity of rot (300 %), while Aspergillus flavus displayed the lowest (100%). This is in contrast with the mild pathogenicity (++) recorded by Amusa et al. [18]. There was no significant difference p>0.05 between the weight losses and percentage severity of rots amongst the test molds with both methods except in the case of D. alata var abana mmee tested with Fusarium solani which displayed high percentage rot severity of 450.00 by the method of Ogbo and Agu as against 153.85 displayed by the method of Okigbo and Nmeka (2005). | ||||||
|
Conclusion
| ||||||
|
The comparative pathogenicity tests carried out by the method of Okigbo and Nmeka, (2005) and a method developed in our laboratory helped determine the actual pathogens from the opportunistic pathogens and the non-pathogens, while the former method demonstrated that all the thirteen initial isolates were pathogenic, the later proved that only seven out of the thirteen isolates were actually pathogenic. It was, however, concluded that since molds were amylolytic in nature the opportunistic pathogens may have capitalized on the wounds created on the tubers to elicit disease symptoms. Therefore, pathogenicity was built on the ability of isolates to be able to colonize, penetrate and elicit the original disease symptom it was isolated from. This knowledge would be helpful to yam pathologists to find out more accurate pathogenicity testing methods which would be useful in the reduction of post-harvest loss of yams. | ||||||
|
Acknowledgements
| ||||||
|
We acknowledge the efforts of the entire Laboratory Staff of Microbiology Department, NnamdiAzikiwe University for helping out with the preparation of the culture media for the Mycological Analyses of the various fungi used in this study. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Frank Chukwunwike Ogbo – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Kingsley Chukwuebuka Agu – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2015 Frank Chukwunwike Ogbo et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|